Methods
The Methods section, often formally called Materials and Methods or Experimental Methods in the body of a journal article or poster, is where scientists explain exactly how their project was conducted.
Writing this section can seem daunting at first because it requires very technical knowledge of your project. However, it often ends up being the easiest section to write, because you’re only writing about things you’ve actually done, usually over and over again, and are therefore very familiar with.
Because your results (and therefore your discussion and introduction) are based on your methods, the Methods section should usually be the first section of your paper that you write. Luckily, you can start writing it as you progress through your project. Plus, your methods will often be based on established methods in your lab, so you can frequently use previously published Methods sections as models for your own.
[bg_faq_start]
Audience and purpose
As we hinted at above, the purpose of the Methods section is to describe how the procedures were conducted exactly. As you will see when you analyze move structures later in this lesson, this means everything from listing the chemicals and other materials you used to explaining what types of mathematical analyses you conducted. Others should be able to replicate your results using the methods you have provided. As you write your Methods, continually ask yourself: “Is this enough information to allow my reader to successfully replicate my experiment?” If not, you’re probably forgetting key pieces of information.
Addressing this purpose helps your audience, other experts in your field, to develop and compare similar procedures and to reproduce and verify your results. Unlike in your Introduction, your Methods section has a consistently expert audience. This means that your tone should stay consistently formal, your content technical, and your voice passive.
Perhaps most important to addressing your expert audience in the Methods section is a highly scientific level of detail. This means not only including all of the necessary information, but also knowing what pieces of information are superfluous. Ask yourself: “Without this detail, could my reader still successfully replicate my experiment?” If so, you might not need to include it. Examples of unnecessary information include:
- Safety information
- Use of everyday equipment (e.g. beakers, pipettes)
- Masses and volumes of reagents used to make solutions
- Details about omitted data
[bg_faq_start]
Practice exerciseRe-write the following lab manual instructions into a format appropriate for a journal article Methods section. Omit any necessary details.
[bg_faq_start] Possible solutions
[bg_faq_end] [bg_faq_end] |
[bg_faq_end]
[bg_faq_start]
Style and conventions
The style of the Methods section can be one of the hardest for new scientific writers to get comfortable with. Read the following excerpts to better familiarize yourself with the way a Methods section might sound.
| Adapted from Wilson et al. (2015):
2.2. MAT gene discovery and MAT locus structure Contigs showing hits with an E-value of 10-5 or less for the MAT and MAT locus-associated genes were subjected to gene prediction using the web-based AUGUSTUS gene prediction tool.33 These contigs were annotated using the AUGUSTUS GFF output file and the ‘‘annotate with GFF/GTF/GVF file’’ function on CLC Main Workbench 6.8 (CLC Bio, Denmark). Gene identity was confirmed using these sequences as queries in an NCBI BLASTn search within the non-redundant nucleotide database.34 Any MAT genes identified in H. omanensis were used as queries in local tBLASTn searches against the H. moniliformis genome.
|
| Adapted from Trabelsi, Szönyi, and Geribaldi (2001):
4.2. Alkylation of dialkyl malonate with 2-perfluoroalkyl-1-iodo ethane in presence of NaH To a suspension of 50 × 10-3 mol of NaH (80% in oil) in dry 100 ml of THF, 0.1 mol of dialkyl malonate was added dropwise over 30 min. After cessation of H2 evolution the mixture was stirred at room temperature for 20 min. To this solution, 50 × 10-3 mol of 2-perfluoroalkyl- 1-iodo ethane was added dropwise, and the mixture was refluxed for 18 h. The solvent was evaporated; the residue was dissolved in 100 ml of Et2O. The organic phase was washed with 30 ml of H2O and dried (Na2SO4). The Et2O layer was concentrated under reduced pressure and the resulting oil purified by distillation under reduced pressure to yield products 4a–f. |
The material is so technical that to non-experts, it can seem extremely dense and dry. So students will often try to write their Methods in a way they think sounds more user-friendly, like:
Once the vacuum pressure was reduced, the layer of Et2O was concentrated, leaving only an oil remaining. This oil was distilled in order to purify it, which again was performed at a reduced pressure, a process which provided us with products 4a–f.
This student-written version, though perhaps more story-like, is wordier and more confusing. The story of your methods is told most accurately through expert, formal, and concise language.
The easiest way to become more comfortable with these techniques and the way Methods sections are constructed in general is to read as many journal articles as you have time to read. The more you read, and then write to try to emulate those articles, the more you will find yourself naturally conforming to the accepted style.
Avoiding ordinal language
Ordinal language, or words that indicate order or position in a series (such as “next,” “then,” or “first”), should be avoided in journal articles. Although it’s tempting to use these words because they sound like they make your methods “flow” better, they are often unnecessary and detract from conciseness. Consider the following two statements:
a. “The powder was first slowly added to the solution and then stirred continuously for 10 minutes. Next, the temperature was raised 1 ˚C per min until it reached 39 ˚C, after which the bottle was capped and finally frozen at -20 ˚C overnight.” (43 words)
b. “The powder was slowly added to the solution and stirred continuously for 10 minutes. The temperature was raised 1 ˚C per min to 39 ˚C. The bottle was capped and frozen at -20 ˚C overnight.” (35 words)
Statement (b) manages to clearly communicate the order of events without using ordinal language and is therefore more concise while maintaining all meaning.
Sometimes, of course, these words might be necessary in order for your methods to be fully understood. If it makes your writing more concise, don’t feel like you absolutely cannot use ordinal language. “After,” for example, is a common word used in Methods sections.
Using formal scientific language
Throughout your methods section, be especially sure to use appropriate scientific language. One of the easiest mistakes to make is to accidentally give too much detail about a common scientific procedure (likely because you don’t yet know how routine it is). What is considered “routine” will, of course, greatly depend on your audience, but it can be guaranteed that lots of the information you might include in a lab report will be superfluous in a journal article-style paper.
For example,
Variances calculated with an F-test were compared to determine in which respective directions the differences in average values would lie
can be written more concisely and professionally by instead saying:
Bidirectional hypotheses were tested by analysis of variance (ANOVA).
Likewise, beginning scientific writers must be vigilant in differentiating between shorthand and accepted scientific abbreviations. Shorthand is a great tool in our scientific notebooks to save us time and therefore accomplish more in the lab, but it is easy to become so familiar with a certain abbreviation that we forget that it’s not universally accepted or entirely professional.
Some types of shorthand are obviously unprofessional, such as writing “w/” instead of “with.” But what about words that seem to toe the line of professionalism?
| Abbreviations to avoid in professional Methods sections | |
| Correct use | Incorrect use |
| “bp: 212 ºF” | “boiling point: 212 º Farenheit” |
| “deionized water” | “DI water” |
| “NaCl was added” | “salt was added” |
| “MW = 33 kDa” | “molecular weight = 33 kilodaltons” |
| “percent yield” | “% yeild” |
| “reaction” | “rxn” |
| sample names like “Lower Elwha River” | “3A” or other nondescript sample names |
| “the sample was washed 3x” | “the sample was washed three times” |
| “week,” “month” or “year” | “wk” “mo” or “yr” |
If you’re ever unsure of the type of language appropriate to describe one of your methods, remember that the literature is a reliable tool for getting ideas for how to phrase something.
[bg_faq_start]
Practice exerciseRewrite the following sentences to better follow the style of the Methods section. Pay particular attention to the level of formality and the use of ordinal language.
[bg_faq_start] Possible solutions
[bg_faq_end] [bg_faq_end] |
Reporting instrument details
In your Methods section, you must always describe the instrumentation you use. As you may have already experienced, scientific apparatus range from being very general to being very specialized and uncommon. The amount of detail you need to provide for each type of instrument with similarly vary. Furthermore, the decision to include a certain piece of information can also depend on your professor or journal of choice—your audience and purpose, as usual, are important factors.
Some instruments are so simple that they don’t require anything more than a quick mention. This would be something like an everyday mercury thermometer or a tabletop centrifuge. To help you decide what types of apparatus warrant further explanation, ask yourself:
- Does the instrument itself influence the quality or nature of the data?
- Is it a larger instrument or one that costs more than $5000?
- Does your analysis require high sensitivity, so that not just anything you could purchase would work?
- Does it have any special settings that could influence a result or the quality of the data?
If the answer to any of theses questions is yes, then you most likely will need to include some more information about the product that just its name.
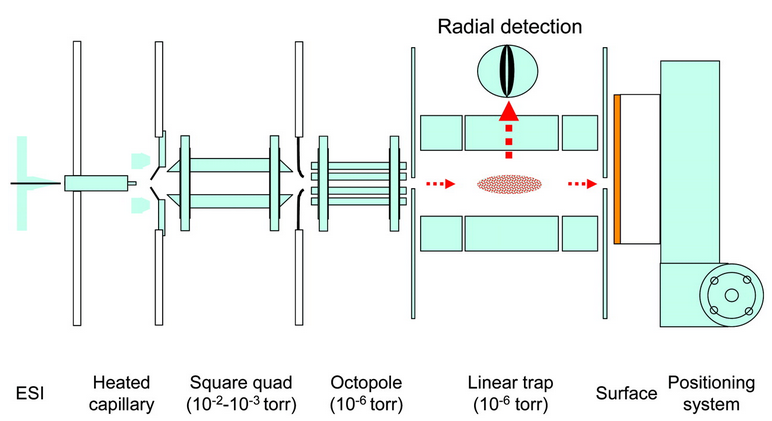
For an idea of the types of apparatus that require a bit more imformation, read the following example, which mentions one technical yet general instrument (with a modification) and one custom instrument.
| Adapted from Ouyang et al. (2003):
The experiments were performed with two instruments: a commercial SSQ-710C (Thermo Finnigan, San Jose, California) quadrupole mass spectrometer that was modified by addition of a surface and its surface moving stage … |
Notice that because a quadrupole mass spectrometer is a common and commercially available apparatus, the authors only needed to state the instrument’s company name and model number.
| Some common instrumentation used in the sciences | |
|
|
Because these instruments are common, you do not need to explain how they work. However you do need to:
- Write out the entire instrument name (such as “atomic force microscope) at first use. If you will refer to the instrument often, you should follow the name with the instrument’s abbreviation in parentheses and henceforth only refer to it by such.
- Provide the model number and company name, preferably in parentheses after the instrument name or abbreviation.
- Report the operational parameters under which you used the instrument.
Operational parameters are the specifications you set your instrument to when you used it. These can often be overlooked by novice writers but are of critical importance to others trying to understand your project in detail. One instrument can be used a multitude of ways by various users to achieve the same task. The parameters you use can greatly affect the outcome of your experiment, so to make your results reproducible, they must be described in detail.
For example, when using HPLC, you must report numerous operational parameters. In the following excerpt, a full sub-section was devoted to this task:
| Adapted from Ma et al. (2006)
A Waters-2695 Alliance HPLC system (Waters Corporation, Milford, MA, USA) equipped with DAD detector at 254 nm was coupled to a Micromass ZQ 2000 electrospray mass spectrometer (Manchester, UK). The Masslynx v. 3.4 software (Micromass) was used for data acquisition and processing. An spherigel C18 analytical column (4.6 mm × 250 mm, 5 μm, Johnsson Dalian, China) was used. The mobile phase system was consisted of A (acetate buffer solution containing of 20 mM ammonium acetate and 1% acetic acid) and B (CH3OH). The linear gradient elution was programmed as follows: 0–5 min, 20–35% B; 5–12 min, 35–100% B; 12–30 min, 100% B; 30–31 min, 100–20% B, 31–35 min, 20% B. The flow rate was set at 1 mL/min while the column temperature was maintained at 30 °C. The injection volume was 20 μL. Only 0.2 mL/min portion of column eluent was delivered into the mass spectrometry by solvent splitting. The ESI-MS spectra were acquired in the positive and negative ion mode. Nitrogen was used as both desolvation gas at a flow rate of 300 L/h and cone gas at a flow rate 50 L/h. The desolvation temperature was set at 250 °C. The ionization source was working at 105 °C. Scan range was 200–850 u. Capillary and cone voltages were 4000 and 30 V for ESI+, and 2500 and 30 V for ESI−, respectively.
The parameters reported are: |
||
|
|
|
And even when just using a telescope, certain parameters must be noted:
Observations of HD 4732 at Okayama Astrophysical Observatory (OAO) were made with the 1.88 m telescope and the High Dispersion Echelle Spectrograph14 from 2004 August to 2012 January. A slit width of the spectrograph was set to 200 μm corresponding to a spectral resolution (R = λ/Δλ) of 67,000 by about 3.3 pixels sampling. For precise radial velocity measurements, we used an iodine absorption cell (I2 cell), which provides a fiducial wavelength reference in a wavelength range of 5000–5800 Å. (Adapted from Sato et al., 2013)
Let’s return to our original example of how to refer to common versus custom instruments. Here is the continuation of the example from Ouyang et al. (2003):
In order to fully describe the instrument, the authors chose to include a diagram. This is common in papers that first describe a custom instrument. Subsequent papers might only highlight the essential features of the apparatus, such as in just this text portion of the example above:
The custom-built instrument29 included facilities for low-energy transfer of trapped, mass-selected ions onto specific spots on an electronically movable target.
A reference to the paper that originally described such an instrument should always be included.
|
[bg_faq_start]
Test yourselfFor each of the following passages, identify: (A) the instrument(s) used (B) if it is (1) an everyday tool, (2) a commercially-available, specialized instrument, or (3) a custom-build apparatus. What clues did you use to make this decision? (C) the parameters given
[bg_faq_start] Solutions
[bg_faq_end] [bg_faq_end] |
[bg_faq_end]
[bg_faq_start]
Organization
Move structure
The goal of a methods section is to describe how a project was conducted in sufficient detail that a fellow expert could replicate the work; thus, the moves used in the methods section are very specific. However, because of the nature of different types of projects, the move structure of this section may look very different between disciplines.
Note that the Methods section is only applicable to an experimental article. In theoretical articles, such as those published in many physics journals, the introduction is often directly followed by an explanation of the theory. Physics papers in general are the least likely to follow a general methods move structure; individual journals should be consulted for preferences. In many geology articles, a section detailing the location of the experiment or observations is sometimes included before the methods section.
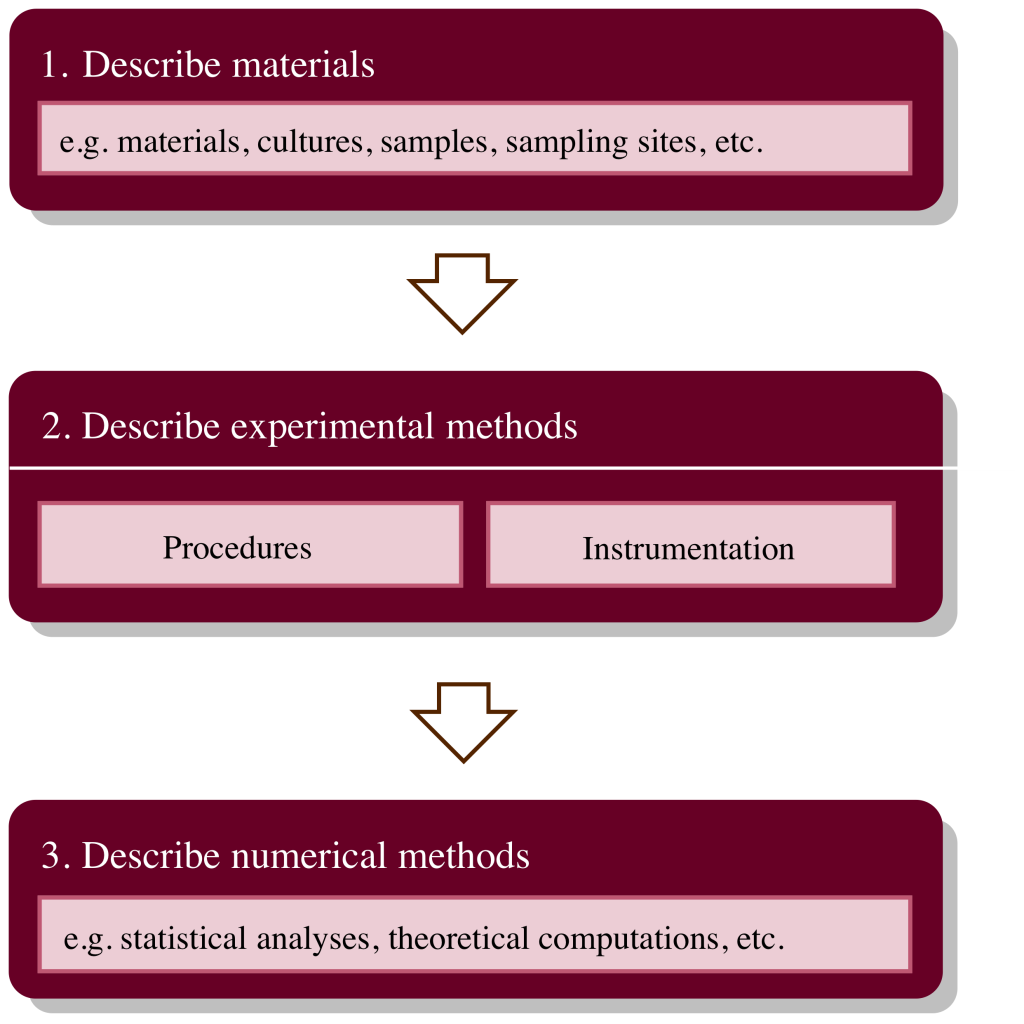
Here is a common move structure for an experimental Methods section:
Each major move can be divided further into sub-moves through the use of subheadings that categorized the different types of materials, instrumentation, or numerical methods, as appropriate. Alternatively, it may sometimes be appropriate to group your methods such that you repeat sets of Moves 1, 2, and 3 for each part of your project. This can sometimes make your methods section more clear and is often used in organic chemistry articles.
1. Describe materials. In the first move, you should mostly list (in full sentences!) the items you used to conduct your experiments. In an experimental physics paper, the experimental setup/design can sometimes be considered Move 1. Remember that you need to provide as much information as necessary for an expert to repeat your project–this means that you will probably need to provide every detail you have about an organism you are studying but minimal details about a general reagent you used to sterilize your glassware. Depending on the project, you might only need a single sentence to complete Move 1 or have no need for Move 1 at all, in which case your Methods section can start with Move 2.
| Examples of Move 1 Adapted from Tripathi, Kaur, and Jyoti (2015): 2.1. Chemicals used Cadmium acetate (Cd(CH3COO)2) and benzene were purchased from Qualigens fine chemicals. Sodium sulfide (anhydrous purified) and ammonia were purchased from Merck Specialities Pvt. Ltd. Polyvinyl pyrrolidone (PVP) and polystyrene (PS) were obtained from Sigma Aldrich. Deionized water was used in all experiments and all of the other chemicals were of analytical grade and used as purchased without any further purification. Adapted from Ghiraldini, Crispim, and Mello (2013): Animals Female NOD/Unib and BALB/cAnUnib mice were obtained from the Multidisciplinary Center of Biological Investigation of the University of Campinas. The animals were reared under standard controlled conditions, fed extruded chow (Nuvital, Colombo, Brazil), and provided water ad libitum. |
2. Describe experimental methods. This move is used to describe how results were obtained. This might involve both experimental procedures and specific instrumentation, which should be described in the order they were conducted in your study. Experimental procedures include field sample collection, synthetic procedures, modeling parameters, quality assurance/quality control (QA/QC) procedures, and anything else that was used to obtain the results of your project. Like in Move 1, the move structure here is highly variable across disciplines.
3. Describe numerical methods. In this move, you should describe any statistical or otherwise mathematical manipulations your conducted on your data to obtain reported results. If none were conducted, this does not need to be stated explicitly, and you can omit Move 3 altogether. If the data analysis of each part of your procedure was conducted differently, it may be appropriate to follow each sub-section of Move 2 with Move 3, then repeat as needed. The key is that manipulation of your results should follow the collection of the results, just as they do in your project.
|
Examples of Move 3 Adapted from Ghiraldini, Crispim, and Mello (2013): All statistical analyses were performed with Minitab 12 software (Minitab, State College, PA). Data comparison between the diabetic animals and their controls from five independent sets of experiments was performed by means of the analysis of variance (ANOVA) test, and for pairwise comparison Student’s t test was used. p < 0.05 was considered the critical level for rejection of the null hypothesis. Adapted from Campos et al. (2015): Data for elastic modulus and flexural strength were analyzed using one-way ANOVA/Tukey’s test. For both tests, the global significance level was 5%. Regression analyzes were performed to |
Remember that the construction of your methods section relies completely on how the work itself was conducted. This means that the order of your sub-moves should imitate the order of your procedures, which is why Move 1 (describe materials) precedes Move 2 (describe procedures) and Move 2 precedes Move 3 (describe numerical methods): usually, you must obtain your materials before using them for experiments, after which you will analyze your data with numerical methods. Furthermore, if, for example, your procedure only involved calculations, you probably won’t have a “describe materials” move, and your experimental procedures will be your numerical methods.
| In an article by Johannsen et al. (2015), the methods section is called “Computational Details” and only includes details about how and why they used certain mathematical manipulations. |
[bg_faq_start]
Org-5: Test yourselfEach of the following sentences are taken out of context from journal article abstracts. Identify the move associated with each statement.
[bg_faq_start] Solutions
[bg_faq_end] |
[bg_faq_end]