|
|
|
|
|
|
My research involves looking deep within the body without making an incision. This is similar to ultrasound imaging, except I am interested in using light instead of sound. Light provides superior resolution, allowing you to see details on the cellular level. How can you see through the body? If you have ever looked at a flashlight pressed under your hand, you have witnessed light traveling through thick tissue. Biomedical imaging entails using lasers, nonlinear optics, and other clever tools to extract images from beneath the surface of biological tissue. With applications in cancer research, nanoparticle characterization, fiber optic endoscopes, and in vivo imaging, these efforts together will provide access to a breathtaking array of unlabeled biological structures. By combining concepts in condensed matter physics, electromagnetism, quantum mechanics, optics, and biology, this area of research is ideal for undergraduate learning and an enrichment of their understanding of physics.
Multiphoton Microscopy
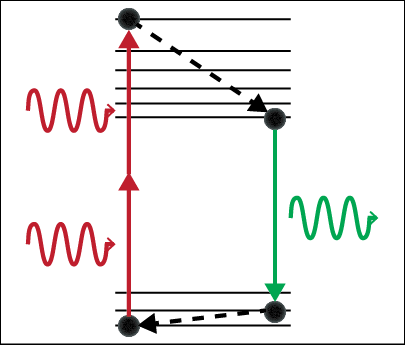
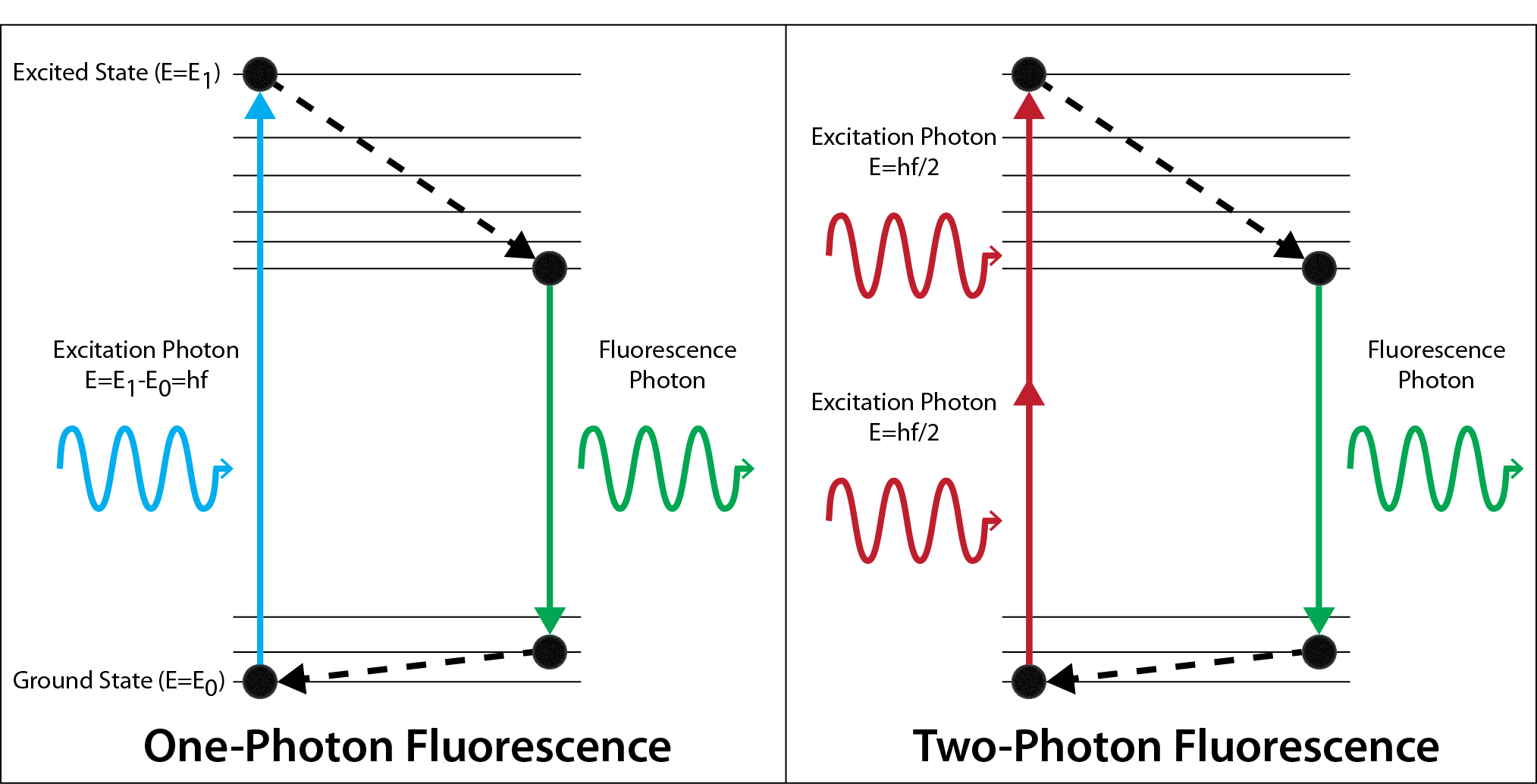
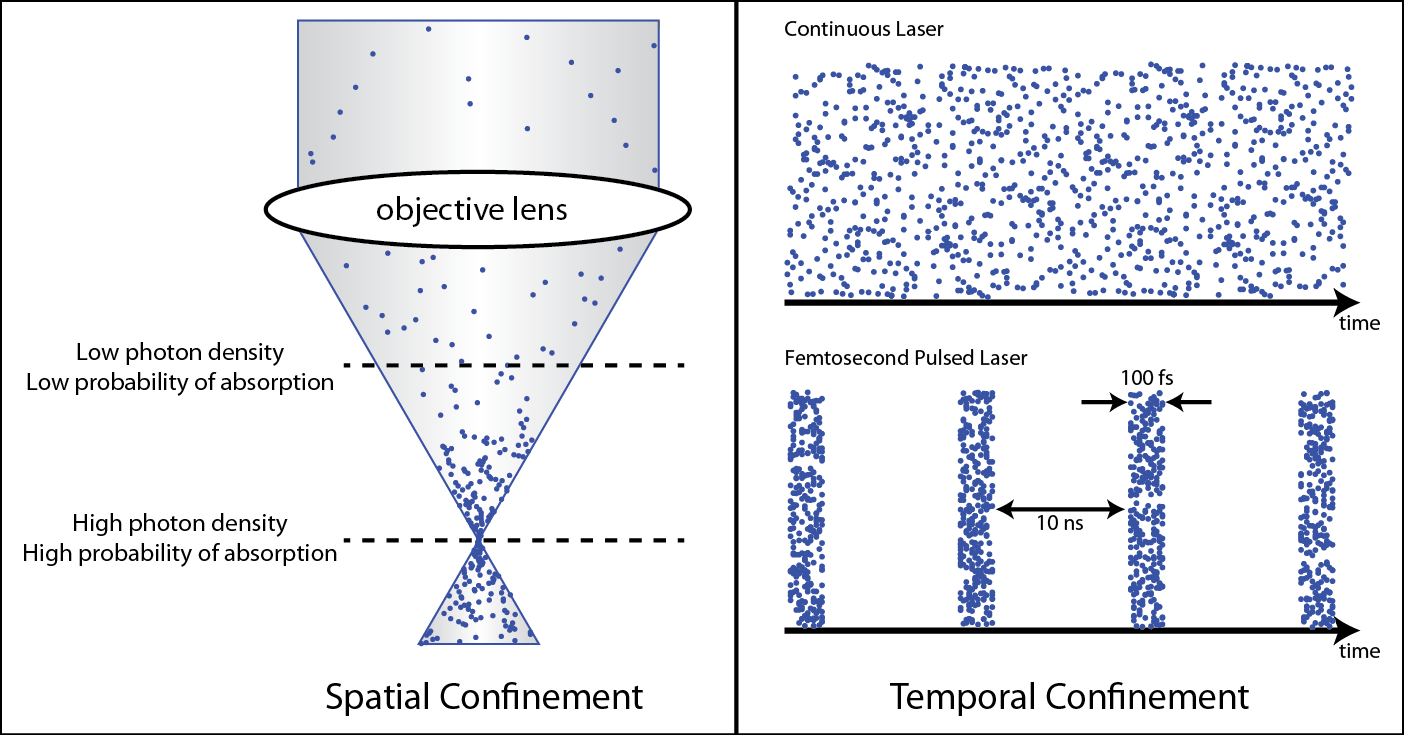
Two-photon microscopy can selectively image cells from one particular depth, taking an effective optical “slice.” Similar to MRI, this technique acquires an image from each depth and stacks these images together to create a 3D image of the biological tissue. Two-photon absorption occurs when a fluorescent dye is excited by two photons that each carry half the energy required for absorption. This is statistically unlikely to happen because the two photons must arrive at the same place at the same time in order to be absorbed. This ensures that the signal must come from the focus of an objective lens, allowing for imaging deep below the surface of scattering biological samples.
In standard one-photon fluorescence microscopy, only one photon is required to excite a dye molecule to an excited state. With two-photon fluorescence, two photons of half the energy can also excite the dye molecule, but the two photons must arrive at the same molecule at the same time. We utilize ultrashort pulsed lasers to ensure the photons arrive at the same time. In addition, a high numerical aperture objective lens is used to focus the photons to the smallest possible volume in space.
Simultaneous Spatial and Temporal Focusing
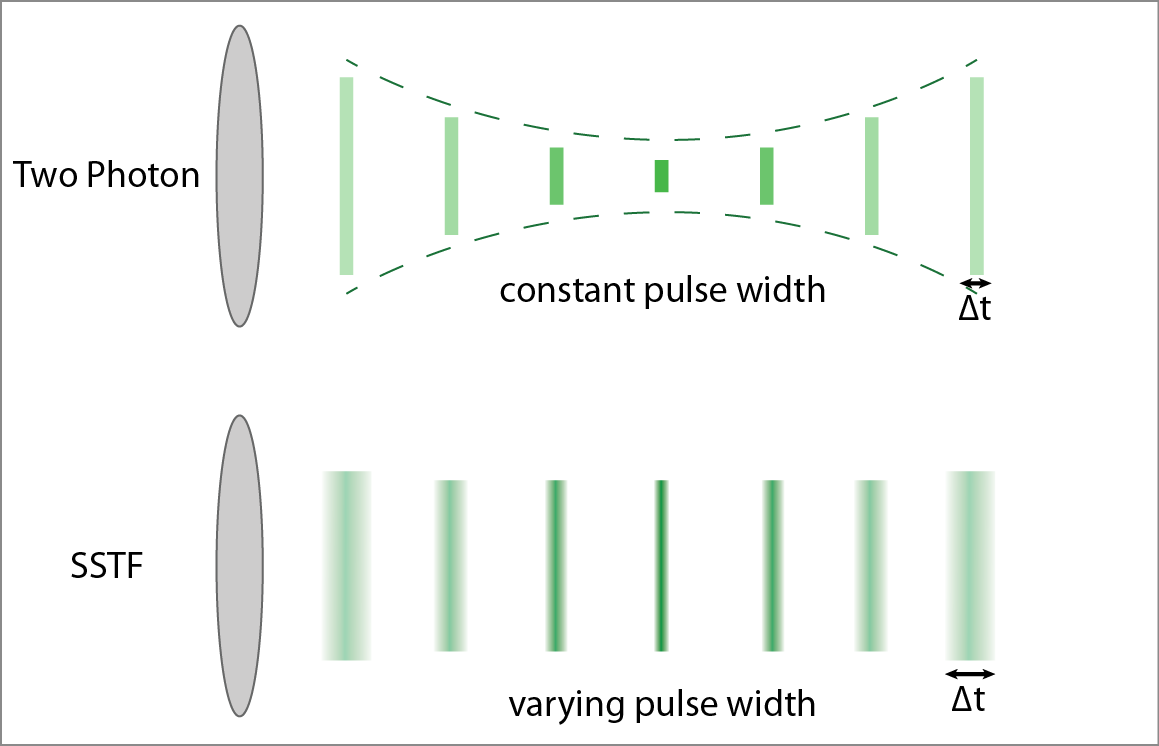
Simultaneous spatial and temporal focusing (SSTF) can improve the speed of two-photon microscopy through wide-field illumination. The shorter the pulse width of the excitation beam, the more likely two-photon absorption will occur. SSTF creates optical sectioning in multiphoton fluorescence imaging by varying the excitation pulse width with position, with the shortest pulse width at the geometric focus of the lens. This ensures maximum two-photon excitation at the focus and reduces background excitation of the biological tissue.
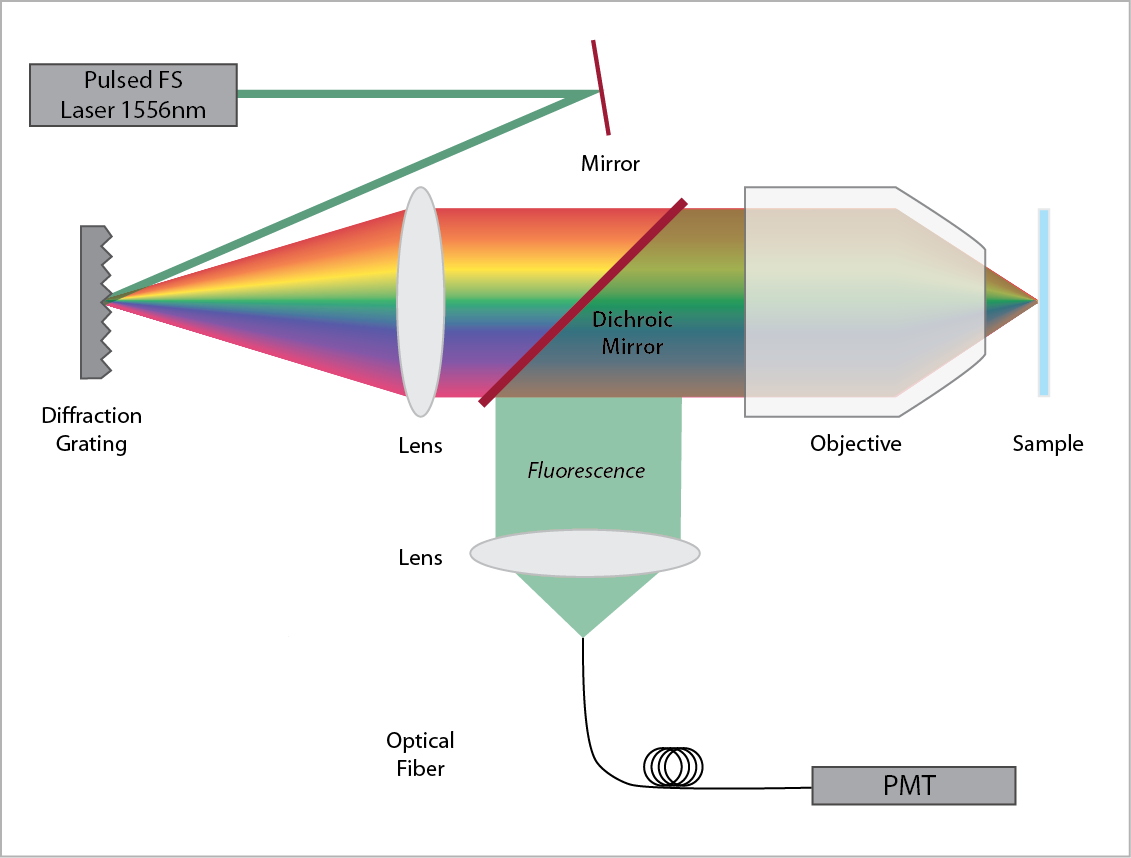
SSTF is implemented by imaging a diffraction grating onto the sample. Because the different frequencies of the laser pulse are spread out in space, a short pulse cannot be formed except at the focus.
Photothermal Imaging
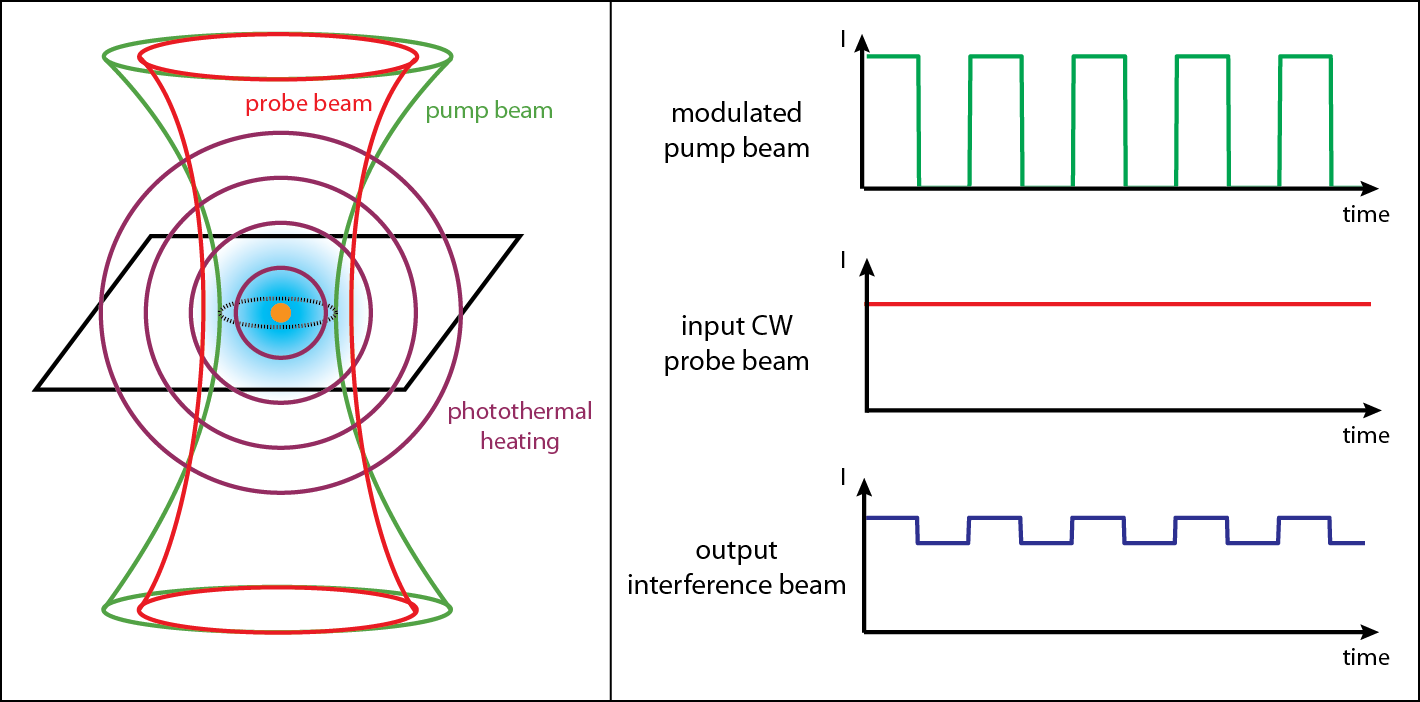
The aim of this project is to enable a regular microscope to see nanoscale absorbing structures. Usually, a biological sample is stained with a fluorescent dye. This project investigates ways to image objects that absorb light without using dyes through a phenomenon called photothermal imaging. Light absorption leads to photothermal heating of the surrounding medium, and the resulting change in refractive index is detected using a pump-probe experiment. This technique can image any object that absorbs light and heats up, including gold nanoparticles and cellular mitochondria. In a biological sample, these absorbing structures can be heated deep beneath the surface through a multiphoton process, allowing for the detection and treatment of cancer and other abnormalities without making an incision.
Because the photothermal effect is weak, lock-in detection of the probe beam must be performed. We heat up the sample using a strongly absorbed pump beam, and we detect changes in the surrounding refractive index using a weakly absorbed probe beam. For sensitive detection, we modulate the pump beam, which causes the photothermally heated region to vary in time. By using a constant power probe beam, we can confirm the presence of the photothermal effect if the outgoing probe beam also becomes modulated at the same frequency.
Nonlinear Optical Harmonic Imaging of Nanoparticles
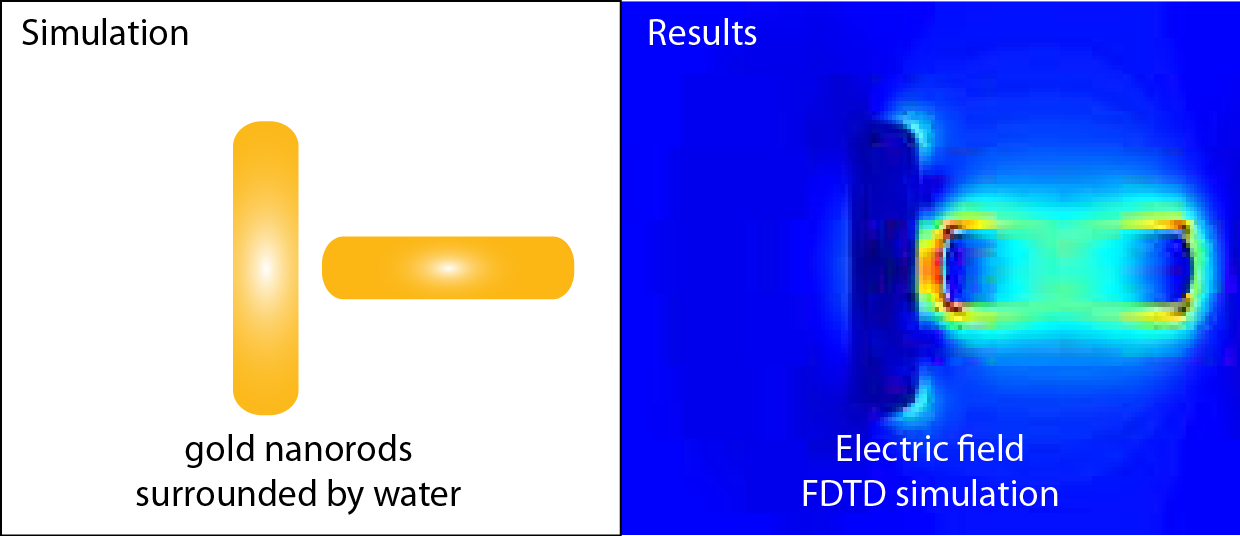
Through a combination of total internal reflection darkfield microscopy and nonlinear optical microscopy and spectroscopy, we survey the second harmonic generation, third harmonic generation, and two-photon luminescence signals from self-assembled gold nanorod structures. The nonlinear optical measurements are made using an ultrafast near-infrared laser whose second and third harmonics are resonant with the gold nanorods’ longitudinal and transverse plasmons, respectively.
Using theoretical simulations, we investigate the effect of gold nanorods’ size, orientation, and coupling modes along with the polarization of incident excitation light on the second- and third-harmonic generation (THG) signal. We obtained theoretical results based on finite-difference time-domain (FDTD) simulations.
Optical Tweezers
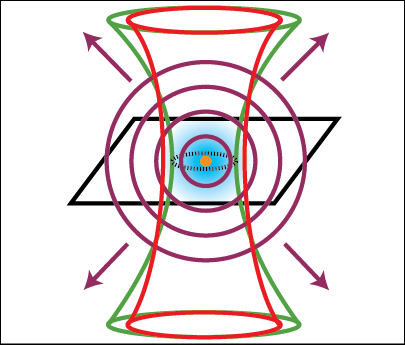
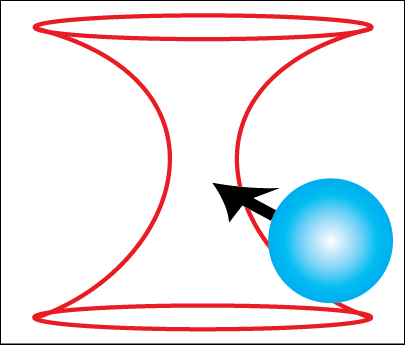
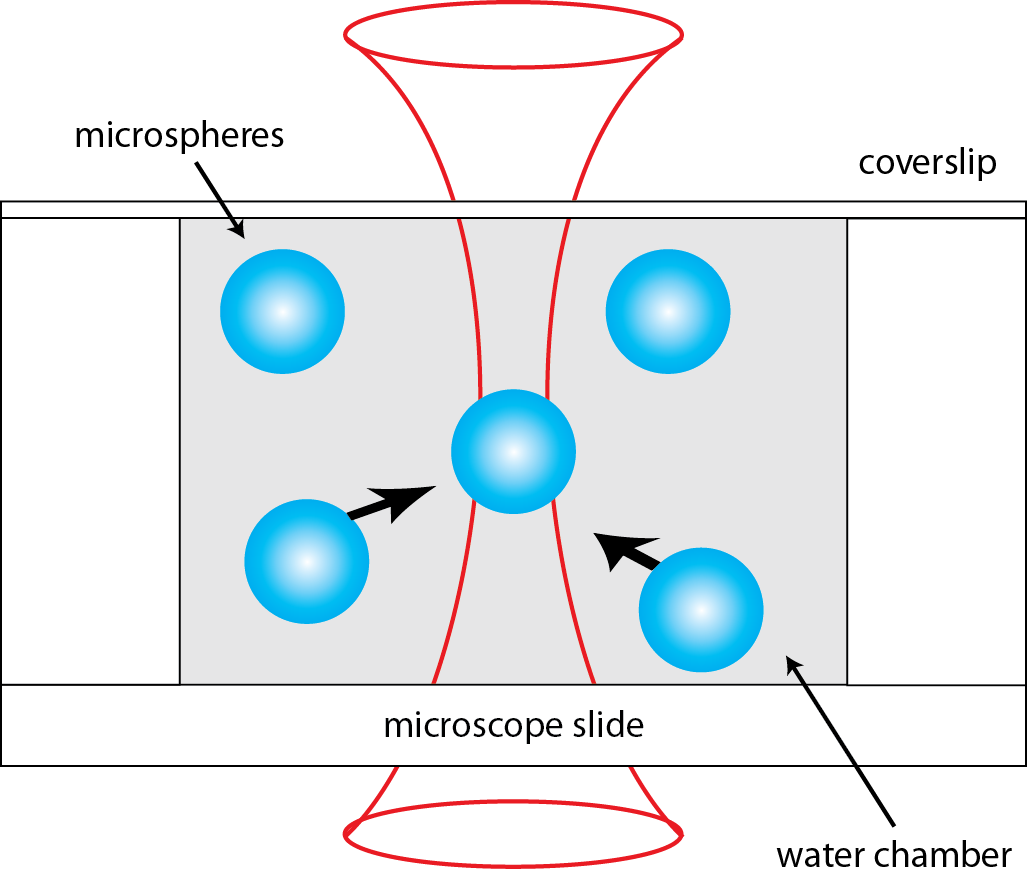
An optical tweezer is an apparatus that uses a light force to physically manipulate microscopic objects, including bacteria, DNA, and polystyrene spheres. My student assembled an optical tweezer which can trap polystyrene microspheres. This experiment consists of the laser trapping beam and a wide-field microscope to observe the trapping. We have shown that the apparatus can trap and manipulate spheres of varying diameters—ranging from 0.5 micrometers microns to 3 microns.
The microspheres are trapped at the focus of a high numerical aperture objective. The tightly focused laser beam creates a steep gradient in electric field, which corresponds to a gradient force pointing inward toward the focus.