Learning goals: follow instructions to complete a chemistry experiment, collect experimental data
Introduction
Models provide a useful way of visualizing the arrangement of electrons in a molecule. As you learned in class, there are several types of structure representations used by the chemist at different times to explain chemical phenomena. Today, we will use 3 of these models to explore chemical structures: Lewis dot structures, a chemical modeling kit, and chemical modeling software.
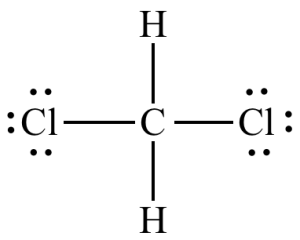
Lewis dot structure
Lewis dot structures allow you to predict molecular arrangements based on the formula. You should be familiar with Lewis dot structures. See an example of a Lewis dot structure on the right.

3D model
Lewis dot structures give no 3D information, so modeling kits can help visualize molecules in 3D. For example, the 3D model makes it obvious why dichloromethane is polar, but this is not obvious looking only at the Lewis dot structure.
ChemDraw is chemical modeling 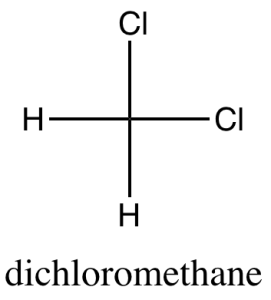 software. It calculates 3D structures and molecular orbitals, which help chemists predict the chemical and physical properties of theoretical molecules. In this lab, you will use ChemDraw to analyze the structures of molecules in 3D.
software. It calculates 3D structures and molecular orbitals, which help chemists predict the chemical and physical properties of theoretical molecules. In this lab, you will use ChemDraw to analyze the structures of molecules in 3D.
Background
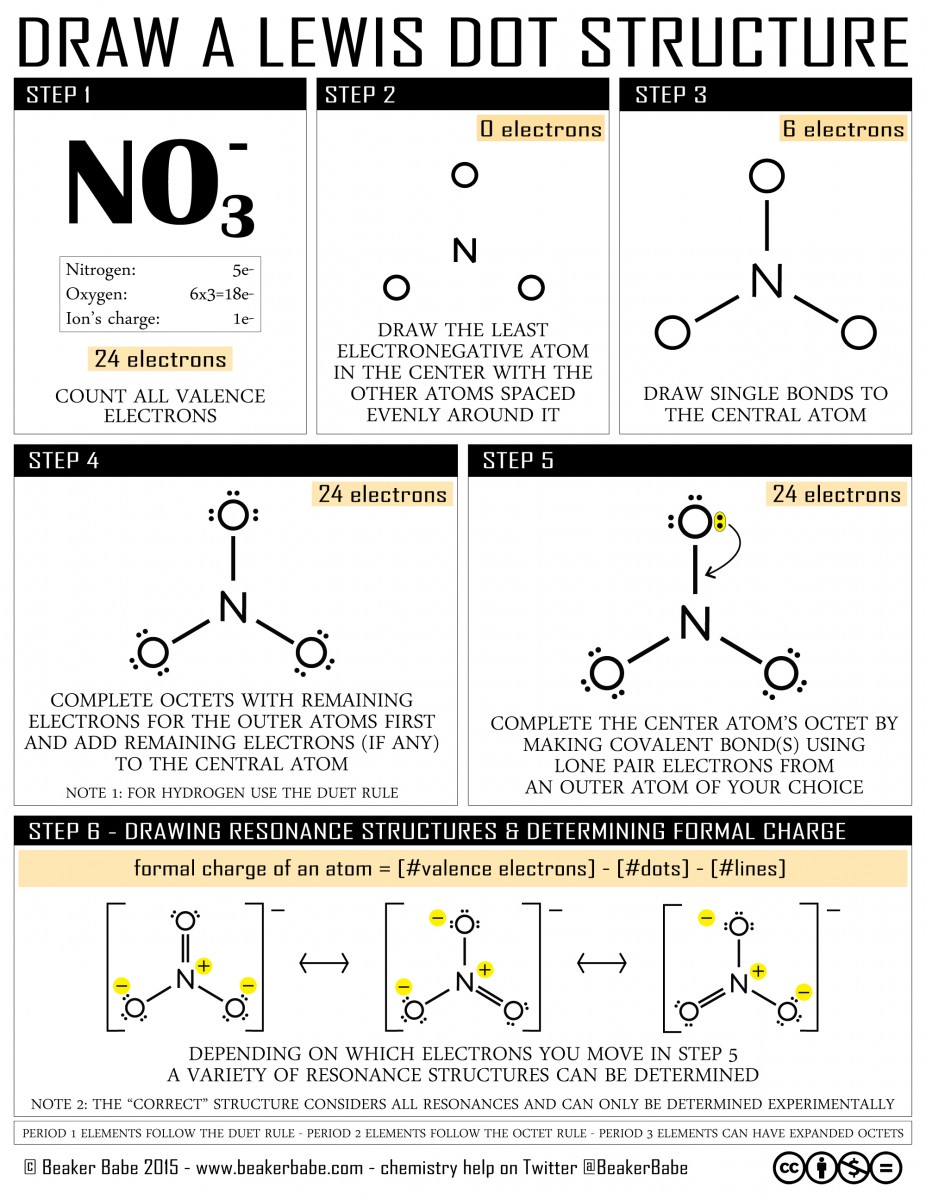
This infographic can help you review Lewis dot structures. Note: step 6 is optional and only applies to some molecules. Also note: below step 6 are some tips on when to follow the octet rule and when the octet rule may have an exception.
Procedure
For each given molecule, first, construct a Lewis dot structure as your pre-lab. Then, in lab, make a 3D model in ChemDraw. Using the model in ChemDraw and your textbook, identify the electron geometry or molecular shape of the central atom (if it is not obvious which is the central atom, just pick one of the central atoms). Answer the discussion questions using the models and the software. Complete the worksheet in lab. Hint: consider revising your Lewis dot structure if it disagrees with the 3D model, but trust your brain more than the computer.
To make a molecular model
- Open Chem3D 15.1
- Click on the white panel to the right of the main window. It is titled “ChemDraw-LiveLink.”
- Click on the “A” icon to type, then type the formula of your molecule of interest. For example, you enter H2O as H2O. When you hit “enter,” the molecule should appear in the big blue window. Note: For more advanced structures or when this method doesn’t produce the structure you were expecting, you will need to type it differently (for example: CH2NH instead of CH3N) or use the tools to draw the molecule by hand. Your instructor and/or TA can show you show you how to do this.
- Optimize the structure by hitting “control-m.” When you do this, the calculated total energy of the model will appear in an output window.
- There are several things you can do to get a better look at the molecule.
- You can click the third button from the left on the top toolbar to rotate the molecule. It looks like a circle with an arrow on it. After you click it, you can use the mouse to rotate the molecule.
- You can click “View” on the main menu, then click “Model Display.” This will present you with many options to change the display of the molecule. For example, “Display Mode” gives you more modes. The “Ball & Stick” mode is most common, but “Wire Frame” is convenient for a very complicated molecule, and “Space Filling” is helpful for visualizing atom size differences.
To measure bond lengths and bond angles on the model
After making the model and optimizing the structure, click “Structure” on the main menu, then “Measurements,” and then select “Generate All Bond Lengths” or “Generate All Bond Angles.”
To measure formal charge of atoms in the model
Hover over the atom of interest. If it has a formal charge, it will say “Formal charge: -1,” for example. It will also display delocalized charges.
To predict the shape of the molecular orbitals
After making the model and optimizing the structure, click “Surfaces” on the main menu, then “Choose Surface,” then “Molecular Orbital.” The HOMO (highest occupied molecular orbital) will automatically be shown, but you can choose another molecular orbital from “Select Molecular Orbital.” You will also see the energies associated with each orbital. The labels are with respect to the HOMO and the LUMO (lowest unoccupied molecular orbital). Notice that the HOMO energy is usually negative, indicating a favorable state, but the LUMO energy is usually positive, indicating an unfavorable state (which is why it is UNOCCUPIED!) The difference between the HOMO and the LUMO energies is called the band gap.
Report
Fill out this worksheet and turn in a paper copy. Optional survey.
